This article was written by César Garcia, researcher at La Hora Maker.
Welcome to the second article in this series on ventilators! As we’ve seen last week, ventilators are critical pieces of infrastructure. They must work reliably for long periods of time without missing a beat. Today we will uncover what are the different phases involved in developing one of these devices. Please, note that this process is a simplified one, based on current circumstances. It usually takes much more time to get one ventilator ready to market.
First stage is the ideation phase. In this initial stage, teams need to decide what technology they will use for their design. One of the most common these days is repurposing an AMBU, by operating it mechanically. There are other alternatives although like pneumatical, based on electro valves, etc, and some of the models approved in Spain involve techniques like High Frequency Jet Ventilation — that is a complete departure from the AMBU models!

Given that the device is going to be used by medical personnel, it’s really important to look at the clinically relevant parameters for these devices. The MIT E-Vent team has done a wonderful job documenting these clinical aspects. You can find the key ventilator specifications to consider on their site.
It’s also worth noting that not all ventilators are meant to work the same. Some of them are better tailored for emergencies, while others are designed to support the patient for longer periods. Mechanical ventilators are covered by several ISO norms like 80601-2-12:2020. Several agencies have made the specifications available for free, to help new initiatives to develop ventilators against COVID-19.
Once you know which approach you would like to take, it’s time to start working on your first functional prototype. Most of the designs will require you to get sensors and valves, as well as basic medical supplies. As per the control unit, we would recommend you to take a look at the Arduino boards better suited to the task in this presentation by Dario Pennisi.
Getting your prototype to pump air is the first step. But you need to control the amount of air in a precise way. Too much-pressurized gas could damage the patient lungs while falling short could suffocate them too. There are two approaches to this issue — some ventilators keep track of the volume of air, while others focus on pressure. To test this, you will need a lung simulator — there are plain simple models to really complex ones. UK’s MHRA offers an extensive test suite for Rapid Manufactured Ventilator Systems (RMVS) for this crisis. You can explore the test at Appendix B.
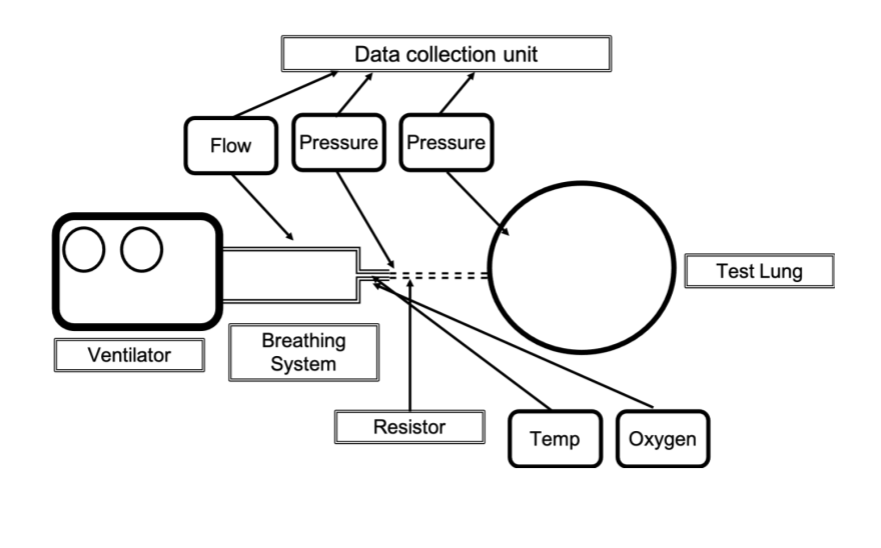
If you are producing ventilators in the UK, this is the main mandatory step right now. In other countries, like Spain, the regulation is a bit more complex — we will focus on those additional steps in the rest of this article.
Once you pass all tests with the simulator, you are required to run clinical tests with animals. As you can imagine, this is not something you can do at your local hackerspace or Fablab. Veterinarians and doctors need to supervise the test, and validate if your device works as expected. Even if you pass some initial tests, you may still need to do more extensive trials. If you plan to produce a non-emergency ventilator, you might be required to repeat tests on pathological animals.
Let’s say you pass all these tests, what is next step? You need to supply your prototype and manuals to an external lab. The goal is to make a third party verify the device specs in a controlled environment. They will test for Electromagnetic compliance, so that the device doesn’t interfere with external ICU equipment, neither is affected by third party emissions.
Once you have your documentation ready, you can submit it for review for the local regulatory agency (AEMPS in Spain, FDA in the USA), to receive final approval! Does this mean that the device is certified? Not really!
Regular certification doesn’t just focus on the device, but also on the manufacturing methods, facilities, quality control, etc. To produce certain equipment, you need to ensure the environmental conditions at the factory, proper hygienic procedures, etc are maintained.
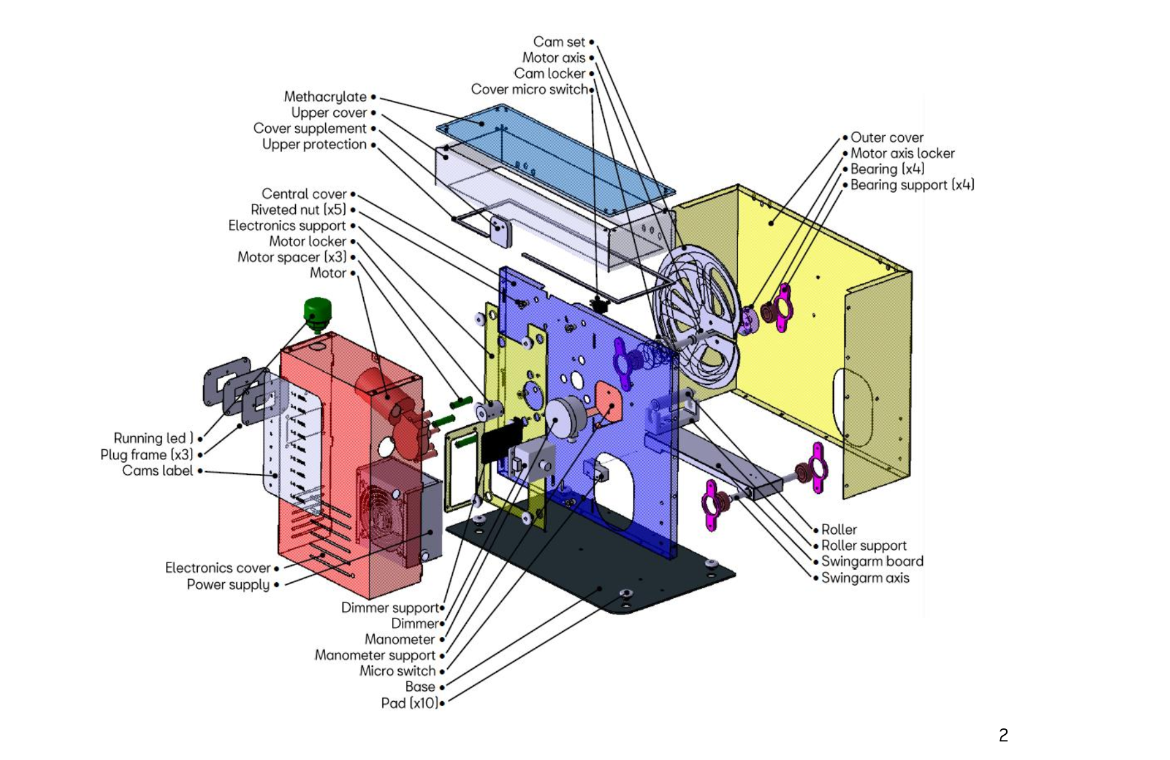
How do you make sure that none of the people assembling or printing is not affected by coronavirus? Most prototypes that have passed all tests have been produced by companies with manufacturing experience. Some projects like Oxygen, offer a maker version and an industrial version, that was manufactured by a car company. In their repository, you can find all documents required to move from prototype to an industrial device!
So, how are these devices going to be deployed? In Spain, they are being used as devices in a clinical trial. Ethical committees in the hospitals would need to approve the trials and set the rules for actual usage. These devices will be used by trained doctors as compassive devices: if no other ventilator is available, they could decide to use them, after getting permission by patients or relatives. These clinical trials would start with a few patients and then scale to larger numbers if required.
In the next episodes, we will explore the stories behind some of these prototypes!
Website: LINK
Schreibe einen Kommentar
Du musst angemeldet sein, um einen Kommentar abzugeben.